SPL Server™

|
|
The first Fully Web-Based, Turn Key
SPL Labeling Compliance Solution
Allow your regulatory operations staff, graphics designers and scientists to collaborate in real time when creating and maintaining labeling content. No new software is required on the end user’s work station. Only a browser is required to access all features and functions including native structured content authoring and document management.
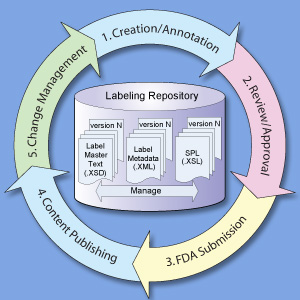
SPL Labeling Process
Designed for Ease of Use
Tier One and Tier Two Validations Are Key
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
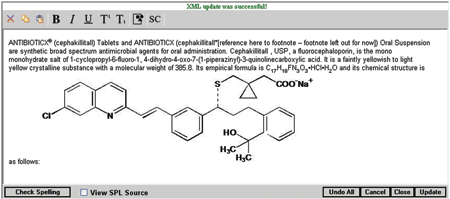
SPL Editor Get real-time validation results with "What You See Is What You Get" (WYSIWYG) editing functionality. Add and delete main sections, sub-sections, paragraphs, tables, ordered lists, unordered lists, and notes all from one easy to use menu. See how your label will be displayed to the public by viewing the SPL using the HL7 standard style sheet. |
|
|
Update SPL No need to train employees on XML with our Rich Text Editor editing functionality. SPL Server makes generating XML simple and seamless. Use standard shortcut keys to make text bold, italic, and underlined or use the SPL Server menu buttons. When you have completed your edits, hit the “Check Spelling” button to check for spelling mistakes. |
|
|
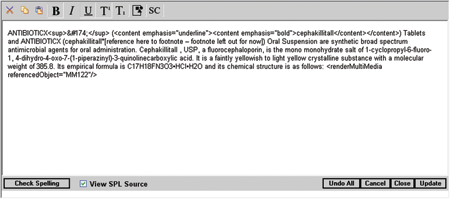
View Source Want to view or edit the SPL source code? Simply check the “View SPL Source” box and let our system convert the text to HL7 schema valid XML. |
|
|
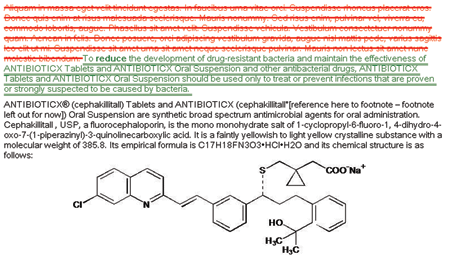
Compare SPL Versions SPL Server takes the guess work out of labeling changes. With our compare functionality you can view differences in any two versions of a label. |
|